
The rate law for this first order rate equation is. We want to know the time required for the reaction to be 75 complete. I am confused about integrated rate reactions and half life problems. So we can obtain our half-life from that formula.
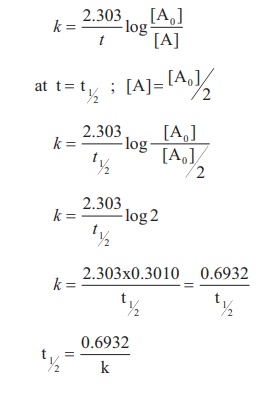
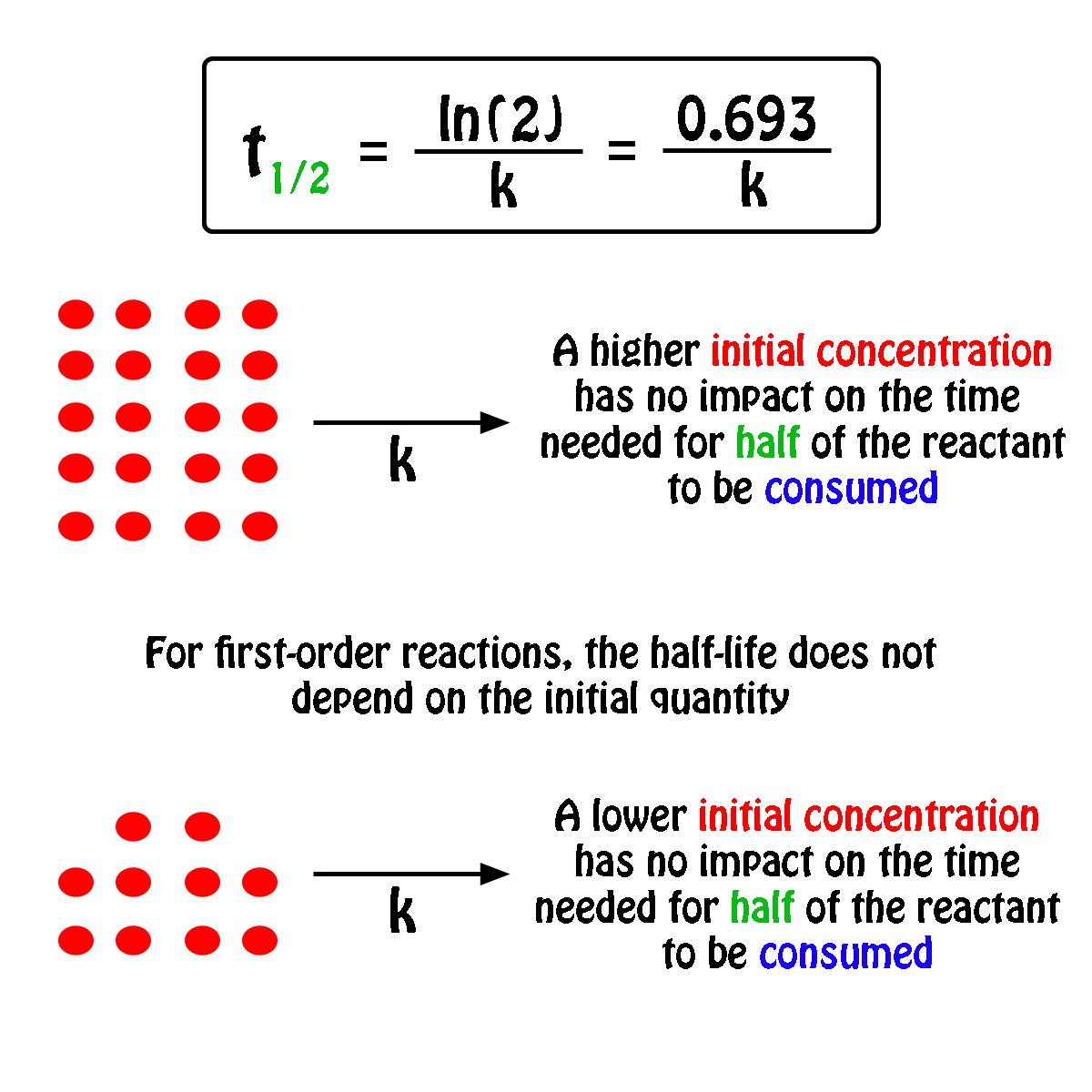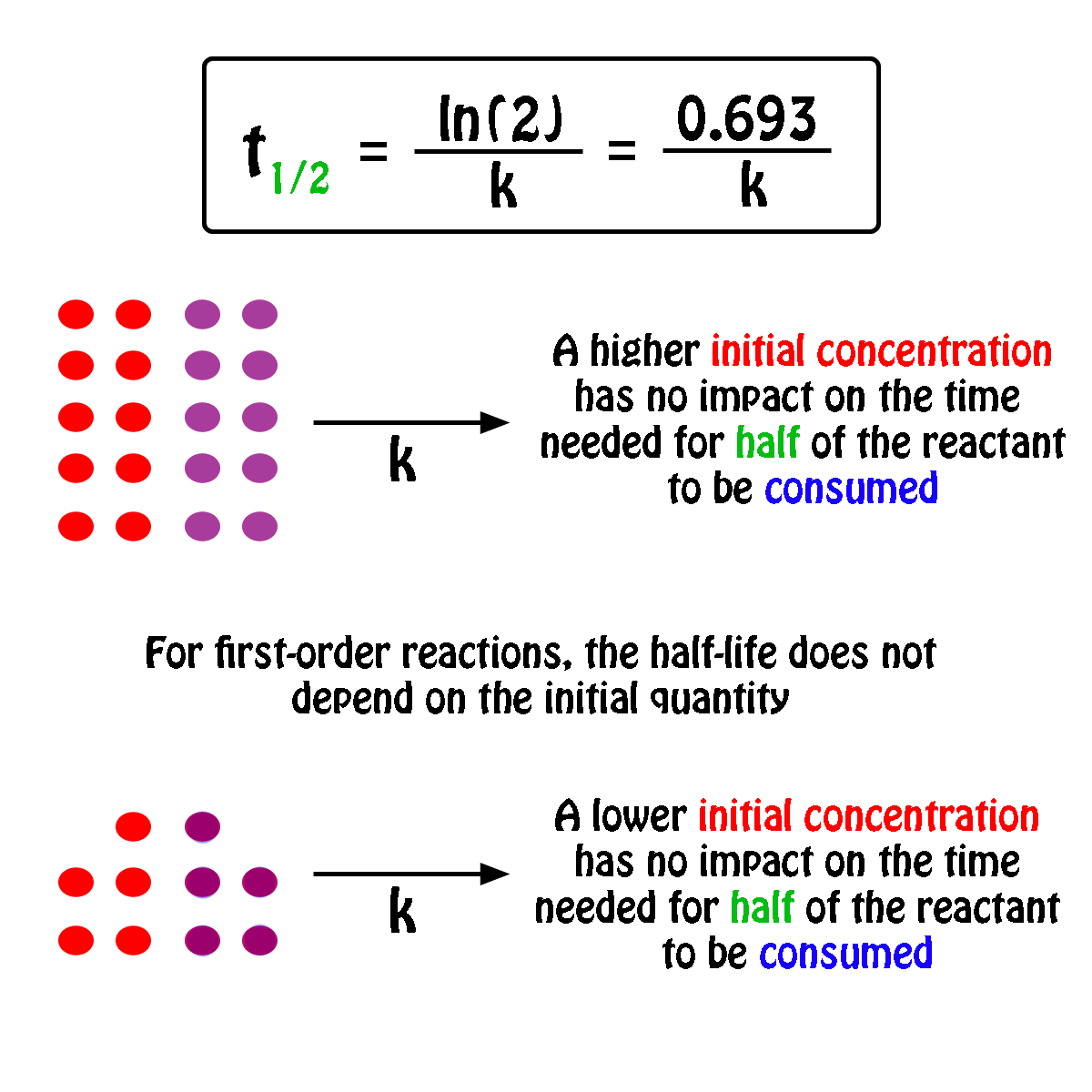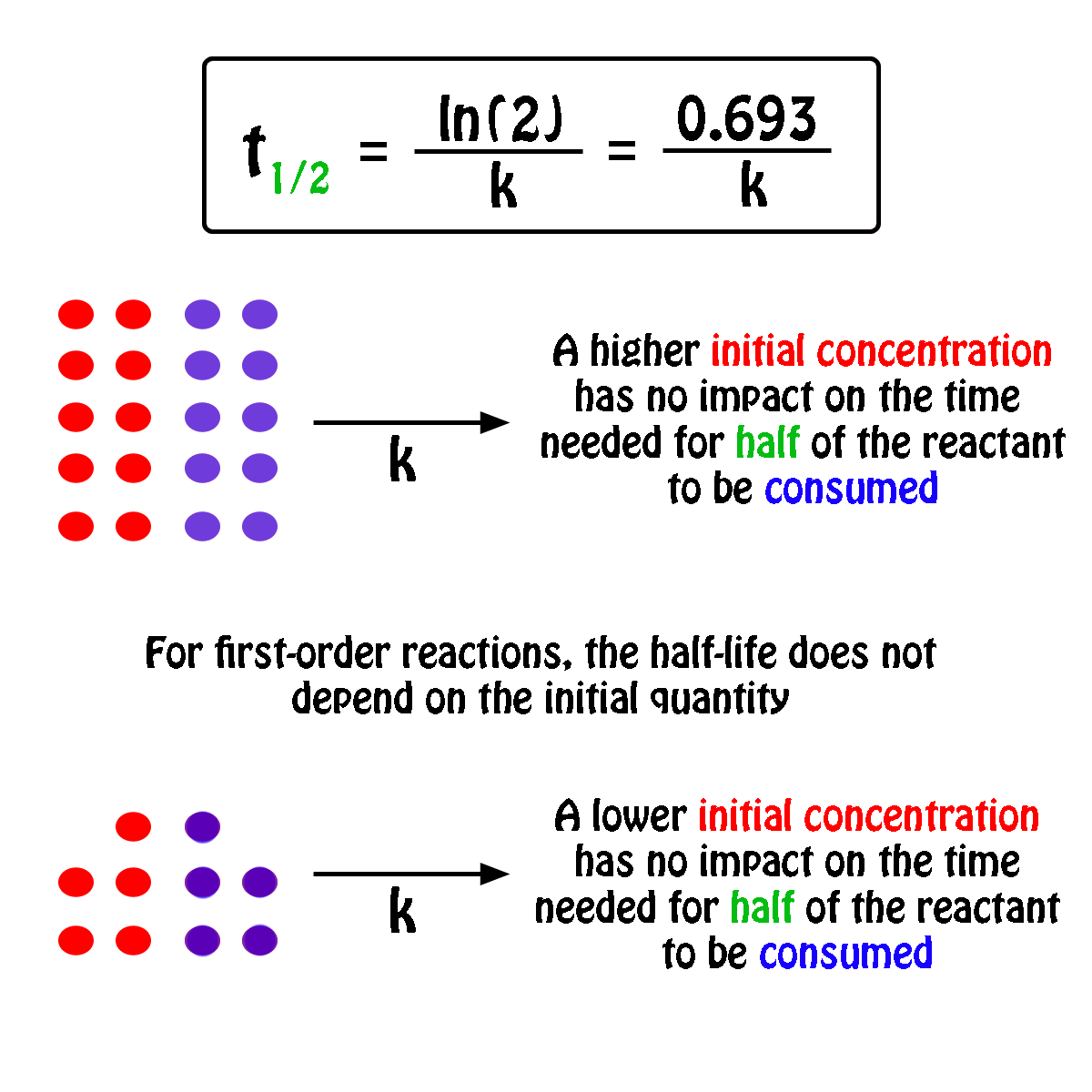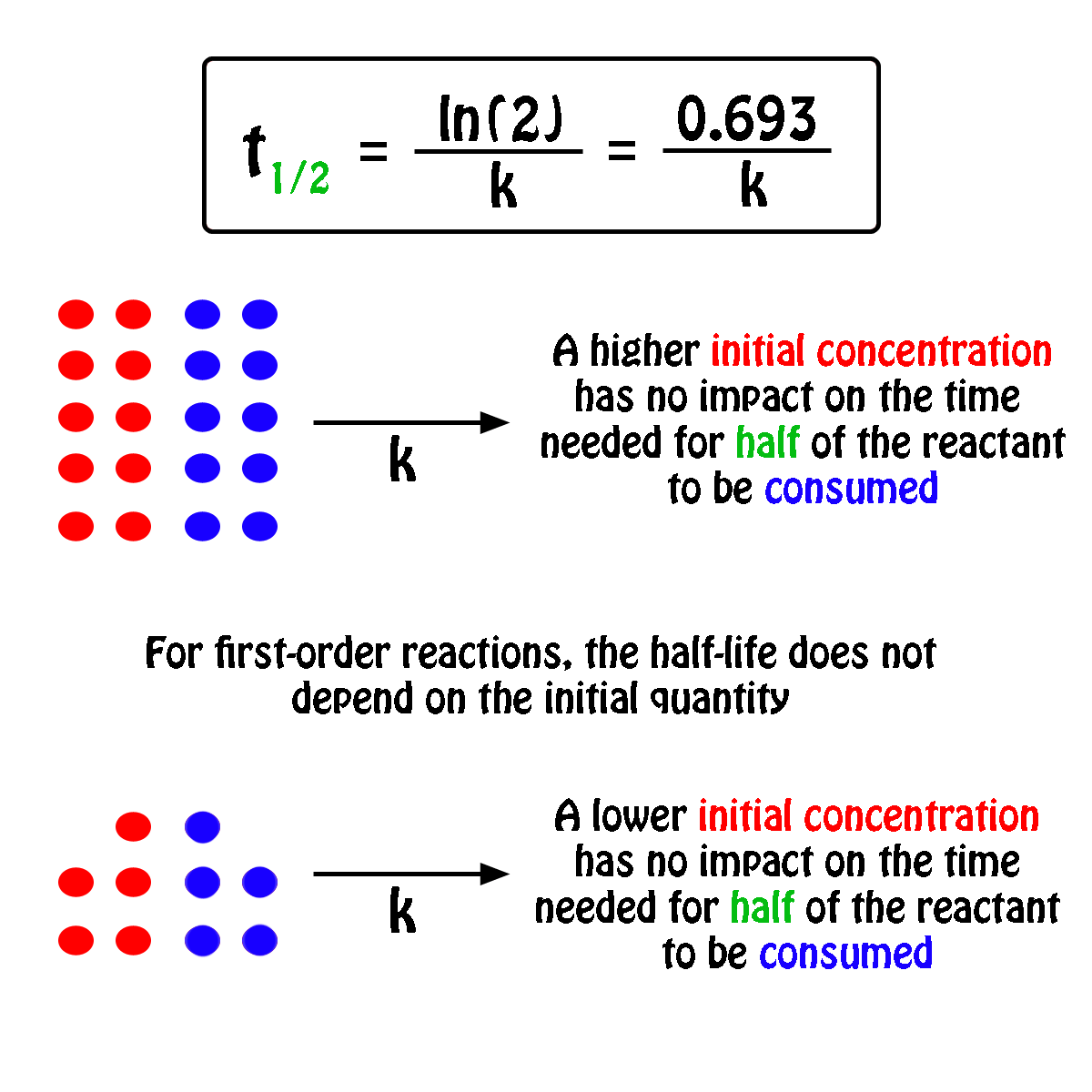
The first part of this derivation, the formula for the rate constant $k$ for the first order reaction, is done in the last part of Chemistry student's answer, and hence I'd not repeat it. Now remember thats our unit for first-order kinetics, always for the rate constant. The half-life of the reaction is 17.0 minutes. Problem 8: The decomposition of hydrogen peroxide is a first-order reaction. Popular Answers (1) You know that in every dynamic 1st order reaction (a reaction with a fixed rate of promotion) we can say that: half life is when N/N01/2. I think what OP need is the derivation of the equation $(1)$ in the question (as indicated in the OP's comment, "an anyone help me with the derivation of the first one"). Since one half-life equals 6.5 hrs, two half-lives would take 13.0 hrs. Half-Life Formula For a zero-order reaction, the mathematical expression that can be employed to determine the half-life is: t1/2 R0/2k For a first-order.
